The Oxygen Window: Mastering Inherent Unsaturation for Efficient Decompression
Introduction: The 'Invisible' Advantage in Decompression
In the world of technical diving, we often talk about "clearing our deco" as if we are simply waiting for a clock to countdown. We watch our computers religiously, tracking our ascent toward that invisible ceiling known as the M-value. But beneath the digital interface of a Shearwater or Ratio computer lies a fascinating physiological phenomenon that acts as a silent partner in every ascent: The Oxygen Window.
Also scientifically referred to as inherent unsaturation, the oxygen window is the physiological "vacuum" created by our own metabolism. It is the delta between the total pressure of gases in our arterial blood and the total pressure of gases in our tissues and venous blood. Understanding this concept is the critical bridge that separates a recreational Nitrox diver from an advanced decompression diver. While a recreational diver uses Nitrox primarily to extend no-decompression limits (NDL), the technical diver uses high-fraction oxygen to "open" this window, aggressively sucking inert gases like nitrogen and helium out of the tissues to shorten decompression times and increase safety margins. 1
By mastering the mechanics of the oxygen window, you move beyond following a series of stop depths; you begin to manage the very engine of gas exchange within your own body.
The Biological Engine: How Metabolism Creates the Window
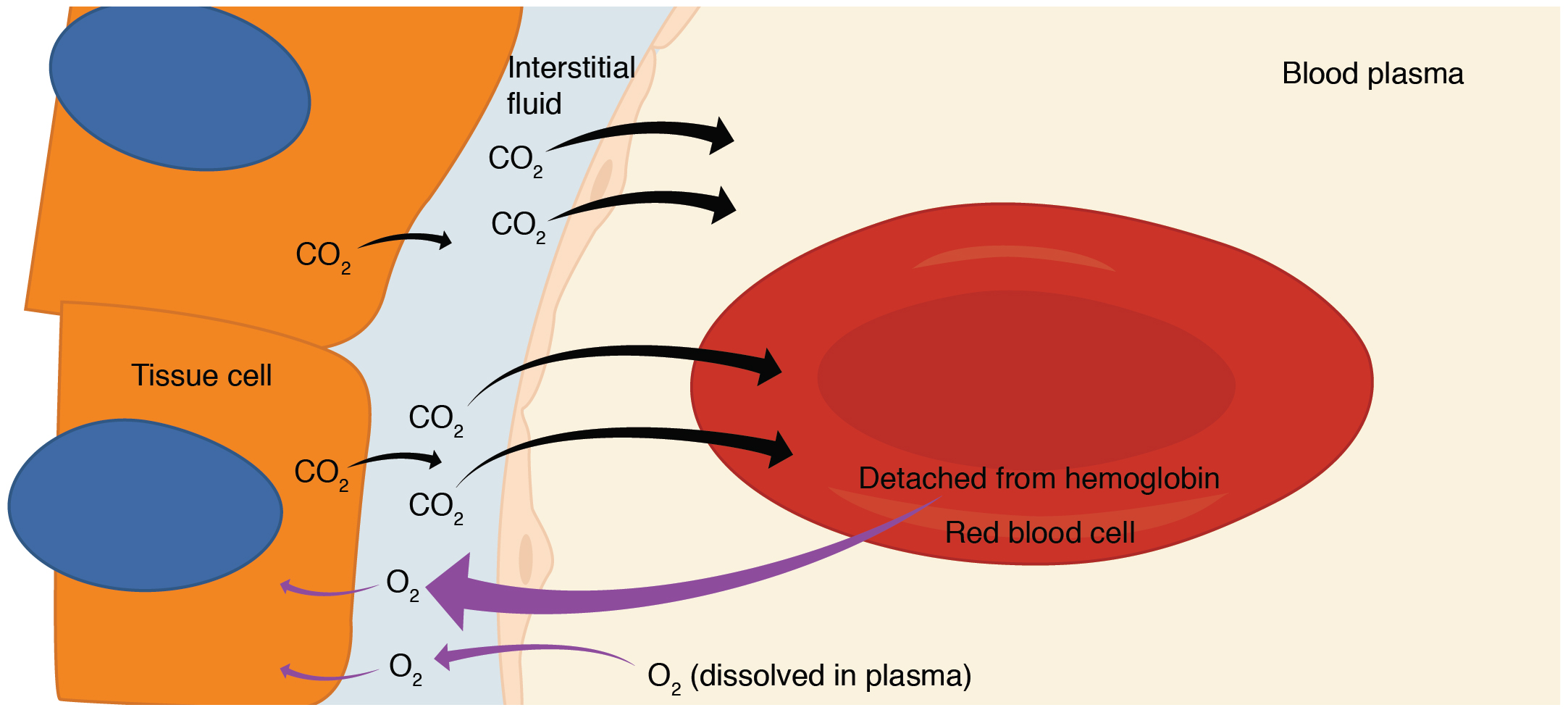
To understand the oxygen window, we must first look at the journey of an oxygen molecule. When you inhale, oxygen moves from the alveoli of your lungs into your arterial blood, where it is primarily bound to hemoglobin. At this stage, the partial pressure of oxygen (PO2) is high. However, as blood reaches the capillaries, the tissues begin to consume that oxygen to fuel the mitochondria—the power plants of our cells. 3
This metabolic process is not a simple one-to-one swap of oxygen for carbon dioxide. Two key factors create the "window":
- The Respiratory Quotient: Humans generally consume more oxygen molecules than they produce carbon dioxide molecules. For every ten molecules of O2 consumed, we typically produce only about eight molecules of CO2 (a ratio of roughly 0.8).
- Solubility Differences: This is the most critical factor. Carbon dioxide is approximately 20 to 25 times more soluble in blood and tissues than oxygen. Because CO2 dissolves so easily into the surrounding fluids, it exerts much less partial pressure than the oxygen it replaced.
The result? The total pressure of all gases dissolved in your venous blood and tissues is significantly lower than the ambient pressure of the environment. This "vacancy" in the total gas tension is the oxygen window. It creates a pressure gap that can be "filled" by inert gases (Nitrogen or Helium) without the total tissue gas tension exceeding ambient pressure, which is the primary driver of bubble formation.
The Physics of Vacancy: Dalton’s Law and Partial Pressure
To visualize this, we must look at Dalton’s Law, which states that the total pressure of a gas mixture is the sum of the partial pressures of its individual components. In our bodies, the total gas tension ($P_{total}$) is the sum of the partial pressures of Oxygen ($PO_2$), Carbon Dioxide ($PCO_2$), Nitrogen ($PN_2$), and Water Vapor ($PH_2O$).
| Gas Component | Arterial Pressure (Approx) | Venous Pressure (Approx) | The "Window" (Delta) |
|---|---|---|---|
| Oxygen (O2) | 95 mmHg | 40 mmHg | -55 mmHg |
| Carbon Di. (CO2) | 40 mmHg | 46 mmHg | +6 mmHg |
| Nitrogen (N2) | 573 mmHg | 573 mmHg | 0 mmHg |
| Water Vapor | 47 mmHg | 47 mmHg | 0 mmHg |
| Total | 755 mmHg | 706 mmHg | -49 mmHg |
Note: Values are simplified for sea-level air breathing to illustrate the concept.
As shown in the table, even at the surface, there is a natural "unsaturation" of about 49 mmHg. In the context of diving, as we descend and ambient pressure increases, these partial pressures scale up. However, because our metabolic rate remains relatively constant (or increases with work), the "drop" in oxygen pressure remains a constant vacuum that we can exploit. 3
Venous blood always has a lower total gas pressure than arterial blood. This is why the oxygen window is the primary mechanism for safe off-gassing; it allows inert gas to move from the tissues into the blood and then to the lungs without reaching the "supersaturation" point where bubbles begin to form. 14
Widening the Gap: How High PO2 Accelerates Off-gassing
The size of the oxygen window is directly proportional to the partial pressure of oxygen you are breathing. This is the secret sauce of technical decompression.
When you switch to a high-O2 "deco gas"—such as EAN50 at 21 meters or 100% O2 at 6 meters—you are doing two things simultaneously:
- Increasing the Gradient: You are reducing the inspired partial pressure of nitrogen ($PiN_2$) to nearly zero (in the case of 100% O2).
- Opening the Window: You are creating a massive drop in total venous gas tension.
When breathing 100% oxygen at 6 meters (1.6 ATA), your arterial $PO_2$ is approximately 1,216 mmHg. While the tissues will consume some of this, the sheer volume of O2 ensures that the venous blood remains highly saturated with O2. However, because there is no nitrogen in the breathing mix, the nitrogen in your tissues is "pushed" into the blood by a massive pressure gradient. 12
This helps overcome the phenomenon of kinetic asymmetry, where nitrogen tends to leave the body slower than it enters. By maximizing the oxygen window, we provide the most efficient "exit ramp" possible for those stubborn inert gas molecules.
The Oxygen Window vs. The Deep Stop Debate
In previous decades, decompression theory leaned heavily toward "Pyle Stops" or deep safety stops, intended to control bubble growth early in the ascent. However, modern research—most notably the NEDU study—has led to a significant shift in how we view the oxygen window in relation to stop depth.
As we discussed in The Deep Stop Debate, staying too deep for too long can actually be counterproductive. When you perform a deep stop, the ambient pressure is still high enough that your "slow tissues" (like fat and bone) may continue to on-gas nitrogen, even while your "fast tissues" (like blood and lungs) are off-gassing. 4
Furthermore, the oxygen window is most "open" when the ratio of oxygen to inert gas is highest. By moving the decompression profile toward shallower depths and using oxygen-rich mixes, we maximize the inherent unsaturation. This is why modern GUE and IANTD profiles often favor a faster ascent from depth to the first gas switch, followed by longer, oxygen-rich stays in the shallow zone (6m to 3m).
Expert Tip:
Deep stops are always safer— actually, modern data suggests that for most technical profiles, shorter deep stops and longer shallow stops on high-O2 mixes provide a more efficient total decompression by keeping the oxygen window wide open.
Constraints and Safety: The Limits of the Window
If a wide oxygen window is good, why not breathe 100% oxygen for the entire dive? The answer lies in the physiological limits of our bodies. Oxygen is a double-edged sword; it is both a metabolic necessity and a potent toxin.
- The Paul Bert Effect (CNS Toxicity): Breathing O2 at high partial pressures (typically above 1.4 ATA during the working phase or 1.6 ATA during deco) can lead to central nervous system convulsions. To stay safe, you must balance the "width" of your oxygen window with the risk of a seizure. You can read more about managing this in our guide to The Paul Bert vs. Lorrain Smith Effect.
- The Lorrain Smith Effect (Pulmonary Toxicity): Long-duration exposures to elevated PO2 can irritate the lungs, leading to a "burning" sensation and reduced vital capacity. This is a major concern for multi-day technical expeditions or very long "rebreather" dives.
- Vasoconstriction: High levels of O2 can cause peripheral blood vessels to constrict. If taken to an extreme, this could actually slow down decompression by reducing blood flow to the very tissues that need to off-gas. This is the law of diminishing returns in action.
Practical Implications for the Advanced Diver
How do you take this theory and apply it to your next dive at the SS Thistlegorm or a deep wall in the Cayman Islands?
1. Optimize Your Gas Switches
Don't just switch because your computer tells you to. Understand that the switch to a higher O2 mix is the moment you "blow the window open." Ensure you are stable, at the correct depth, and have notified your buddy. The goal is to maximize the time spent at the highest safe PO2 for your current depth. 2
2. Manage Your Gradient Factors
Your Gradient Factors (GF) are the tools you use to tell your dive computer how much of the oxygen window you want to use.
- A GF High of 70 or 85 determines how close you get to the M-value at the surface.
- A GF Low of 30 or 50 determines your first stop depth. If you want to utilize the oxygen window more effectively, many modern divers are moving toward higher GF Low settings (e.g., 50/80) to avoid unnecessary on-gassing at depth.
3. Thermal and Metabolic Management
As discussed in Thermodynamics of the Deep, being cold during the decompression phase is a recipe for disaster. Cold causes vasoconstriction, which effectively "shuts the window" by reducing the transport of gas from tissues to the lungs. 3
- Stay Warm: Use heated vests or drysuits to maintain perfusion during deco.
- Hydrate: Dehydration thickens the blood, making gas transport less efficient.
- Light Activity: Gentle movement of the fingers and toes during deco can help maintain circulation without spiking CO2 levels.
Conclusion: Integrating Physiology into Your Dive Profile
The oxygen window is a testament to the incredible complexity of human physiology. It is not just a mathematical quirk of Dalton’s Law; it is a living, breathing process fueled by your metabolism. By understanding that your body creates a natural "vacuum" for inert gases, you can make more informed decisions about your gas choices, your ascent rates, and your deco stops.
As diving theory continues to move away from rigid bubble models and toward physiological-based models, the importance of maintaining a healthy "window" becomes clear. Stay warm, stay hydrated, and respect the limits of oxygen toxicity. When you do, you aren't just following a computer—you're mastering the science of the deep.
Ready to take your theory to the next level? Check out our deep dive on Ascent Rates and Kinetic Energy to see how speed impacts your gas management.
