Beyond Boyle’s Law: How the Joule-Thomson Effect Triggers Regulator Free-Flow

Introduction: The Cold Reality of Gas Expansion
Imagine you are hovering at 30 meters in a freshwater quarry. The visibility is a haunting crystalline green, and the temperature has plummeted to a biting 4°C. Suddenly, the rhythmic hiss of your breathing is replaced by a violent, unrelenting roar. Your second stage begins to deliver a geyser of air, obscuring your vision with a curtain of silver bubbles. You are experiencing a catastrophic regulator free-flow.
For many divers, the immediate instinct is to blame the cold water alone. We are taught early in our training that Boyle’s Law dictates the relationship between pressure and volume 1. While Boyle’s Law explains why our lungs don't collapse and why our tanks provide more gas at the surface than at depth, it is only half the story of regulator performance. The "Ideal Gas Law" assumes that gas molecules are point-masses that do not interact with one another—a convenient fiction that falls apart under the high-pressure environment of a scuba cylinder 2.
To truly understand why a regulator fails in cold water, we must look beyond the simplified physics of open-water courses and explore the Joule-Thomson Effect. This phenomenon describes the relationship between pressure drops and thermal energy, explaining why the very act of breathing can turn your first stage into a block of ice, even if the surrounding water is technically above freezing.
Boyle’s Law vs. Reality: Why the Ideal Gas Law Is Incomplete
In a perfect world, gas behavior is predictable. Boyle’s Law ($P_1V_1 = P_2V_2$) tells us that if temperature remains constant, the product of pressure and volume is also constant 1. When your first stage reduces the high-pressure air from your cylinder (typically 3000 psi) to an intermediate pressure (approximately 140 psi above ambient), Boyle’s Law suggests the volume simply increases to compensate for the drop in pressure.
However, the Ideal Gas Law ignores the reality of molecular physics. As we explored in our deep dive on Van der Waals Equation vs. Ideal Gas Law: Why High-Pressure Fills Don't Follow the Rules, real gas molecules have actual volume and exert attractive forces on one another. These intermolecular forces, known as Van der Waals forces, become significant when gas is highly compressed 2.
The limitation of "Ideal" physics is that it assumes no energy is lost or gained during expansion. In reality, when gas moves from the high-pressure environment of the cylinder through the narrow orifice of the first stage valve, it doesn't just expand; it undergoes a radical thermodynamic shift. Because the process happens so quickly, it is nearly adiabatic—meaning no heat is exchanged with the environment in that split second. Consequently, the energy required for the gas to expand must come from the gas itself.
The Joule-Thomson Effect Explained
The Joule-Thomson (JT) Effect is the temperature change of a real gas when it is forced through a valve or porous plug while kept insulated so that no heat is exchanged with the environment.
At the molecular level, this is a matter of "work." In a high-pressure cylinder, gas molecules are packed tightly together. As they expand into the lower-pressure intermediate hose, they must overcome the attractive Van der Waals forces holding them together 2. Overcoming these forces requires energy. Since the expansion happens too fast to draw heat from the surrounding water, the gas molecules "spend" their own kinetic energy to move apart.
Since temperature is essentially a measurement of average kinetic energy, this loss of energy results in a rapid drop in temperature.
The Cooling Coefficient
Different gases react differently to expansion based on their Joule-Thomson coefficient.
- Air and Nitrox: These gases have a positive JT coefficient at standard diving temperatures, meaning they cool down significantly upon expansion.
- Helium: Under most diving conditions, helium actually has a negative JT coefficient, meaning it could theoretically warm up upon expansion; however, in a Trimix blend, the cooling of the oxygen and nitrogen usually dominates the thermal equation.
| Gas Component | JT Behavior at 20°C | Impact on Regulator |
|---|---|---|
| Nitrogen | Cooling | Significant |
| Oxygen | Cooling | Moderate |
| Helium | Warming (Slight) | Negligible |
| Carbon Dioxide | Strong Cooling | High Risk |
The Mechanics of Icing: From Gas to Solid
When the Joule-Thomson effect is in full swing, the first stage of your regulator becomes a massive thermal sink. The pressure drop from 3000 psi to 140 psi is so violent that the internal gas temperature can plummet by 20°C to 30°C in an instant. This creates two distinct types of icing:
Internal Icing
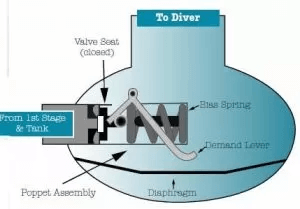
Internal icing is the result of moisture contamination within the breathing gas itself 5. If the air from the compressor was not properly filtered and dried, the water vapor inside the tank will freeze into tiny ice crystals as it passes through the first stage orifice. These crystals, often called an "Ice Plug," can jam the high-pressure seat or the piston, preventing the regulator from closing. This leads to a runaway pressure increase in the intermediate line, eventually forcing the second stage to free-flow as a safety mechanism.
External Icing
External icing occurs even with "dry" gas. The Joule-Thomson effect cools the metal body of the regulator so far below the freezing point that the surrounding water begins to freeze onto the exterior of the first stage 5. If the regulator is a "wet" design (where water enters the spring chamber to provide a pressure reference), ice can form inside the chamber. This ice physically prevents the main spring or diaphragm from moving, locking the valve in the open position and triggering a free-flow.
Gas Composition and Thermal Conductivity
While the JT effect explains the cooling, the gas composition dictates how that cold is managed. This is where we see a fascinating conflict between thermodynamics and gas density.
In our article Thermodynamics of the Deep: Helium’s Thermal Challenge and Your Decompression Budget, we discussed how helium’s high thermal conductivity accelerates heat loss from the diver's body 4. In the context of a regulator, helium’s high conductivity is actually a double-edged sword. While it doesn't contribute much to JT cooling, its ability to transfer heat means it can rapidly pull warmth from the regulator body—or, conversely, allow the cold gas to chill the surrounding metal more efficiently.
Furthermore, high-oxygen mixtures used for decompression require specific material considerations. Oxygen is a highly reactive gas; while it follows similar JT cooling patterns to nitrogen, the presence of any hydrocarbon lubricants in a cold, high-pressure oxygen environment creates a fire risk if the "heat of compression" (the opposite of the JT effect) occurs during cylinder opening.
Environmental Catalysts: When Theory Becomes an Emergency
The transition from a "cold regulator" to a "frozen regulator" is often accelerated by environmental factors and diver behavior.
- Ambient Water Temperature: In water below 10°C, the thermal gradient between the water and the JT-cooled regulator is smaller, meaning the water is less effective at "warming" the regulator back up.
- High Flow Rates: The faster you breathe, the more gas expands through the orifice per minute. This increases the cumulative JT cooling effect. This is why a high Work of Breathing (WOB) or heavy exertion is a primary trigger for free-flow.
- Surface Testing: Testing a regulator in sub-zero air is a recipe for disaster. Air is a poor conductor of heat compared to water 4. Without the high heat capacity of water to buffer the temperature drop, the regulator can freeze internally before you even hit the water 5.
Expert Tip: Cold water also impacts other chemical and mechanical processes. Much like the efficiency drops seen in Scrubber Science: How Pressure and Cold Water Impact CO2 Absorbent Efficiency, your regulator's mechanical tolerances can shift as metals contract at different rates in extreme cold.
Engineering Solutions: Defeating the Freeze
Manufacturers have developed several engineering workarounds to mitigate the Joule-Thomson effect:
- Environmentally Sealed First Stages: By using a secondary diaphragm and a silicone or alcohol-based transmitter, the internal moving parts are kept dry and isolated from the cold ambient water. This prevents external ice from jamming the mechanism.
- Heat Sinks and Ribbed Fins: Many cold-water regulators feature deep ribs on the first stage body. These increase the surface area, maximizing the thermal exchange with the surrounding water to "wick" heat into the cold metal.
- Material Selection: Most high-quality regulators are made from chrome-plated brass. Brass has a high thermal mass and decent conductivity, allowing it to act as a heat reservoir 4.
| Feature | Function | Benefit |
|---|---|---|
| Dry Seal Kit | Isolates moving parts | Prevents external icing |
| Teflon Coating | Reduces ice adhesion | Prevents internal jamming |
| Heat Fins | Increases surface area | Improves heat transfer |
Diver Protocol: Preventing the Joule-Thomson Trap
Understanding the physics allows us to develop better safety protocols for cold-water diving.
Pre-Dive Checklist
- Check Gas Quality: Ensure your air fill comes from a reputable source with well-maintained filtration to minimize internal moisture.
- Avoid Surface Purging: Never purge your regulator in cold air. If you must test it, take a short, gentle breath 5.
- Keep it Dry: Ensure your second stage stays out of the water until you are ready to submerge to prevent "flash freezing" of the orifice.
In-Water Management
- Breath Control: Maintain a slow, steady inhalation rate. This reduces the velocity of the gas and spreads the JT cooling over a longer period.
- Manage WOB: Avoid over-exertion. If you feel your breathing rate increasing, stop and rest to let the regulator's thermal mass recover.
- Managing a Free-Flow: If a free-flow occurs, do not panic. Turn your head so the regulator is slightly higher than the first stage, and "sip" air from the flow. If you are using a dual-manifold system, be prepared to shut down the affected post.
Conclusion: Respecting the Physics of the Deep
The Joule-Thomson effect is a stark reminder that diving is an exercise in applied physics. While Boyle’s Law provides the foundation of our understanding, it is the nuances of real-gas behavior and thermodynamics that dictate the limits of our equipment 12.
A regulator free-flow is rarely a "random" equipment failure; it is a predictable physical reaction to pressure, temperature, and moisture. By choosing environmentally sealed equipment, ensuring high gas quality, and managing our breathing rates, we can mitigate the cooling effects of gas expansion. As advanced divers, looking beyond the surface of basic theory allows us to anticipate these challenges before they become emergencies, ensuring that even in the coldest reaches of the deep, our life-support systems remain rock solid.
Ready to dive deeper into the science of the abyss? Explore our guide on The Oxygen Window: Mastering Inherent Unsaturation for Efficient Decompression to see how gas physics impacts your body long after the dive is over.